Taste is one of the most important sensations for human life, enabling us to perceive different tastes from the diverse range of food available in nature and is a major determinant of our ingestion decisions. These intracellular reactions regulate sensory functions of smell, taste, and vision, and a wide variety of physiological processes such as secretion, neurotransmission, metabolism, cellular differentiation, inflammation and immune responses ( Lagerström and Schiöth, 2008 Rosenbaum et al., 2009 Venkatakrishnan et al., 2013 Ahmad et al., 2015). 
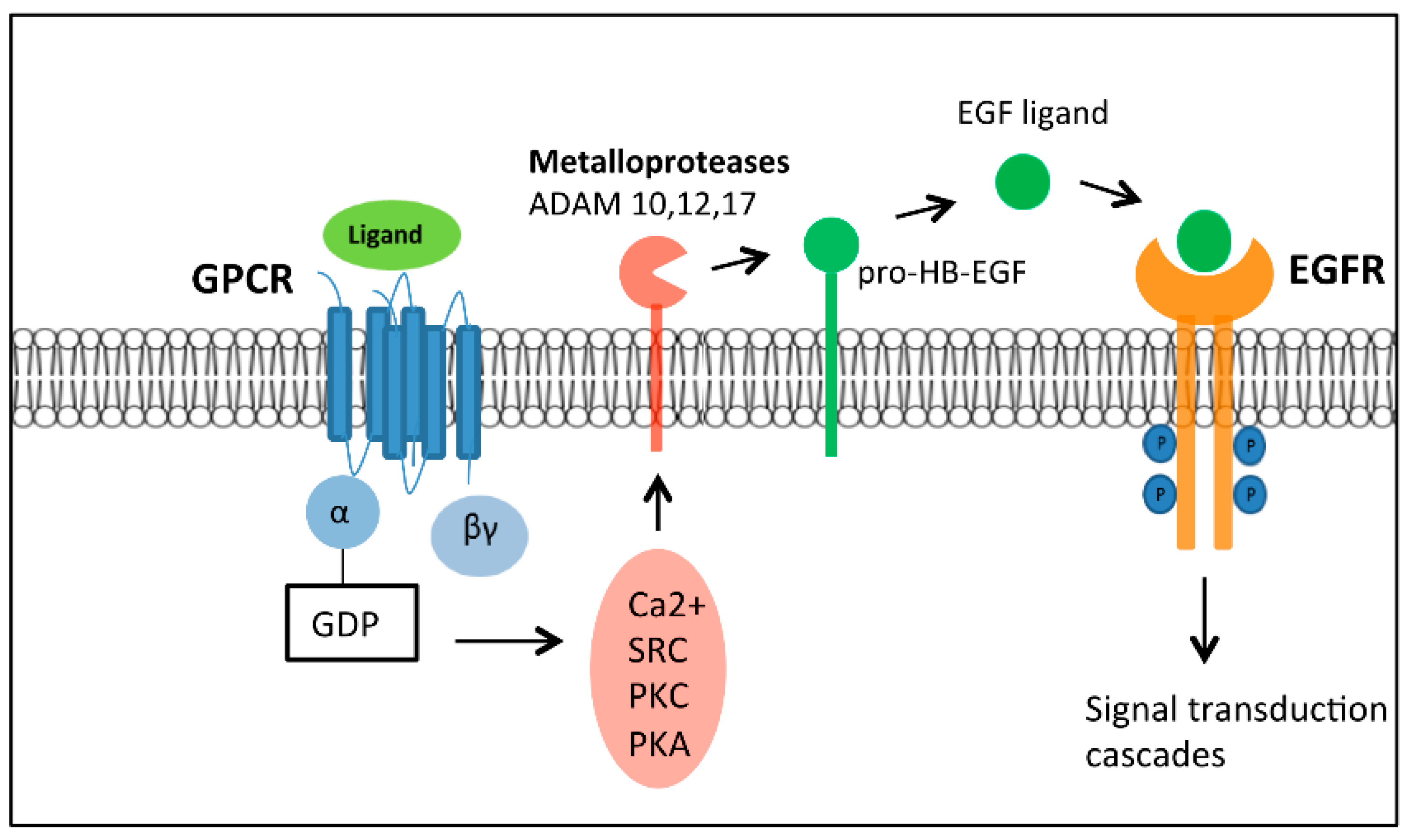
The conformational changes of GPCRs upon ligand binding initiate a series of biochemical reactions within the cell. They are activated by a wide variety of ligands in the form of light energy, lipids, sugars, peptides and proteins ( Billington and Penn, 2003 Schoneberg et al., 2004 Lundstrom, 2009) which convey information from the outside environment into the cell to mediate their corresponding functional responses. G protein-coupled receptors (GPCRs) are the largest and the most diverse group of membrane receptors in eukaryotes. However, the need for more functional studies in a physiological context is apparent and would be enhanced by a crystallized structure of taste receptors for a more complete picture of their pharmacological mechanisms. In addition, studies based on transgenic mice, utilizing either global or conditional knock out strategies have provided insights to taste receptor signal transduction mechanisms and their roles in physiology. In recent years, intensive integrative approaches combining heterologous expression, mutagenesis and homology modeling have together provided insight regarding agonist binding site locations and molecular mechanisms of orthosteric and allosteric modulation. Despite recent advances in our understanding of the relationship between agonist binding and the conformational changes required for receptor activation, several major challenges and questions remain in taste GPCR biology that are discussed in the present review. Tastant binding to the receptor activates downstream secondary messenger pathways leading to depolarization and increased intracellular calcium in TRCs, that in turn innervate the gustatory cortex in the brain. Sweet, umami and kokumi receptors share structural similarities in containing multiple agonist binding sites with pronounced selectivity while most bitter receptors contain a single binding site that is broadly tuned to a diverse array of bitter ligands in a non-selective manner. Type I taste GPCRs (TAS1R) form heterodimeric complexes that function as sweet (TAS1R2/TAS1R3) or umami (TAS1R1/TAS1R3) taste receptors, whereas Type II are monomeric bitter taste receptors or kokumi/calcium-sensing receptors. 
Taste GPCRs reside in specialised taste receptor cells (TRCs) within taste buds. Besides mediating the sensory modalities of olfaction and vision, GPCRs also transduce signals for three basic taste qualities of sweet, umami (savory taste), and bitter, as well as the flavor sensation kokumi. Heterotrimeric G protein-coupled receptors (GPCRs) comprise the largest receptor family in mammals and are responsible for the regulation of most physiological functions. Food Nutrition and Health Team, Food and Bio-based Products Group, AgResearch, Palmerston North, New Zealand. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
